New Analysis of GMP Inspections by MHRA
26/04/2017
Due to the Freedom of Information Act (FOI), the FDA makes their inspection results available to the public. More rarely, however, the public gets informed about inspection results of EU supervisory authorities. The U.K. Medicines & Healthcare Products Regulatory Agency (MHRA) is a positive exception from this rule. Now, the 2016 GMP inspection deficiency data trend has been published by MHRA's GMDP Inspectorate. According to MHRA they have "improved the way of gathering the inspection deficiency data". The new data trending can now allow stakeholders to identify: - The severity and frequency by the EU GMP references
- The overall number of deficiencies by categories: Critical, Major, Other
- The high impact vs. high frequency issues
In 2016, the MHRA has inspected 242 UK manufacturers and performed 82 inspections overseas. In the top-ten list of "critical" and "major" deviations found in all inspected areas, findings regarding the quality system rank first. And there is a new number two: "Sterility Assurance", which wasn't even in the top ten in 2015 (see illustration 1). This is similar to recent FDA findings and the same number one as in the previous year. 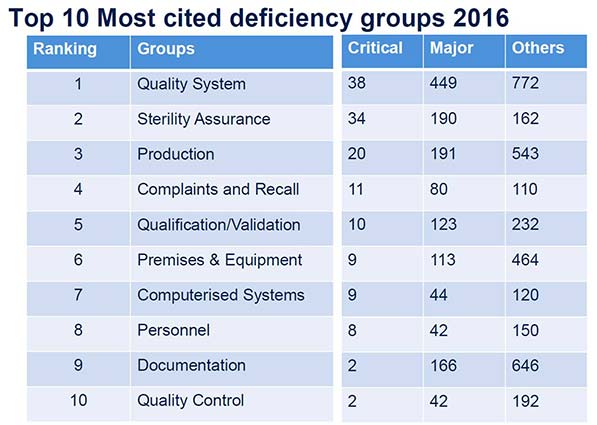 Most of the respective findings in Chapter 1 of the EU-GMP Guidelines (Pharmaceutical Quality System) refer to 1.4 describing the pre-requisites for an appropriate Pharmaceutical Quality System and 1.8 describing the Good Manufacturing Practices needed. Again, a lot of observations were made in the handling of deviations and CAPA, as can be seen in the examples given in the report. But also quite a few deficiencies related to the lack of senior management oversight on effective implementation of pharmaceutical quality system (PQS) and the Product Quality Review (PQR) were made. As in the previous year, the MHRA also focuses on ensuring that updates to EU GMP were captured, reviewed and implemented. The report lists examples for all relevant chapters and annexes of the EU GMP Guidelines, allowing stakeholders to "perform their own assessment against the deficiency findings as part of self-inspection and continuous improvement". When it comes to Annex 16 for example (Certification by a Qualified Person and Batch Release), a Qualified Person (QP) can get a view on what is expected. And this year, some observations were made at the GMP/GDP interface: - "Arrangements for temperature monitoring of air shipments did not justify why data loggers were not required in every pallet."
- "Procedures allowed mean kinetic temperature (MKT) to be used to assess temperature excursions but did not require an investigation to be performed into the cause of the excursion."
- "Several products were routinely imported into the EU from the USA and stored at site for onward export to other third countries, however these were not subject to QP certification before being released for this export supply"
Conference Recommendations | |
|
|



