2 April 2013
Written Confirmations - How is the EU pharmaceutical Industry dealing with that?
From 2 July 2013 on, APIs from Third Countries will be allowed for
importation into the European Union only if the regulatory authority of the
exporting country is confirming that the API was manufactured according EU GMP.
Currently, hardly any other subject is as extensively discussed in the GMP
environment as this so called Written Confirmation.
To find out how this issue is handled in pharmaceutical companies in the EU,
the European QP Association conducted a brief survey amongst its
members beginning of this year. Almost 300 QPs gave answers to the questions.
This is an impressive number which also means that the results are quite
representative.
About 50% of the companies replied that they already tried to get a written
confirmation for APIs manufactured outside the EU. A number which might be
higher now. Only 15% were confident that their company will get the written
confirmation for all APIs imported from outside the EU by 2 July 2013. 35%
didn't think so. For the rest it was too early to say at this stage.
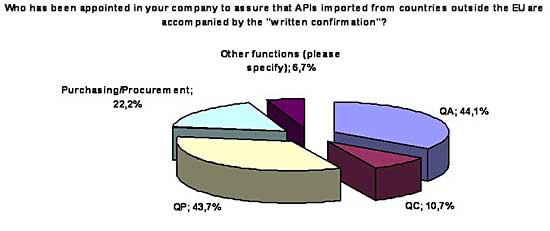
Another question asked who is or will be appointed in the company to assure
that APIs imported from countries outside the EU are accompanied by the Written
Confirmation. It was very interesting to see that in more than 40% of the
companies the Qualified Person (QP) will be directly involved in this process.
With about the same percentage, Quality Assurance functions will be in the
driver seat in other companies; and procurement only in 22% of the companies:
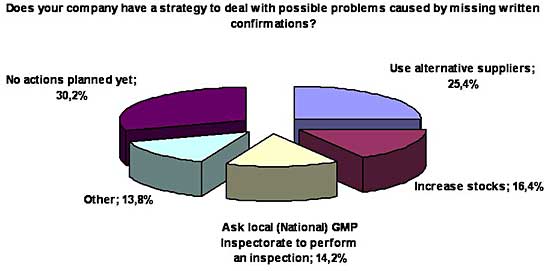
The QP Association also asked about strategies dealing with possible problems
caused by missing written confirmations. It seemed that here was not much
consensus on how to deal with these possible challenges:
There is still a lot of uncertainty in Europe what the future will bring. The
only alternative to a Written Confirmation is the admission to the list of
"Third Countries" with comparable inspection standards. Currently only
Switzerland is on that list and therefore has not to issue Written
Confirmations. Israel's and also Singnapor's request have been rejected (see ECA News
from 20 March 2013). It is unlikely that countries like India and China will
forward a request as their GMP compliance standards are currently not equivalent
with those in the EU. The only alternative for these countries is thus to supply
"Written Confirmations" for every single API. There are currently severe doubts
that this will be possible for all APIs imported into the EU. For that reason
some pharmaceutical companies already started to increase their API
stocks. |



